Atom diagram
The repulsion can be minimized by moving them apart and placing indifferent degenerate subshells.Īll the subshells having a single electron will spin in the same direction, either clockwise or anticlockwise.Įlectrons in molecules, like atoms, begin by occupying lower energy levels. Hund's rule deals with the placement of electrons into decadent orbitals of the same subshells (s, p, d).īonding in s, p, d subshells can not occur until each orbital is occupied by one electron.Īs the electrons are negatively charged, they repel each other. they jump to higher energy levels only when lower levels are filled.Īccording to the principle, an orbital can occupy a maximum of two electrons having an opposite spin. In most cases, the emission begins with an atom stimulated to its higher state either by collision or by absorption of light from the environment.Ībove shows the representation of the Aufbau Principle.Īccording to the principle, in the ground state, the orbitals are filled according to their increasing energies.Įlectrons first occupy the position in lower energy. Thanks to the Grotrian diagram, we can see that light emission and absorption happen at the same wavelengths.Ī molecule or atom travels from a lower energy state to a higher energy state when it absorbs light or collides with another atom or ion that provides sufficient energy. The meaning of the energy level diagram is as follows: It is a technique to remember the order of filling the vacant energy levels. To fill the vacant energy levels, the Aufbau Principle is used. The outermost orbital of lower energy level has higher energy than the consequent orbital of higher energy level. for energy level 3, the arrangement should be 3s<3p<3d.įor energy level 4, the placement of orbitals is 4s<4p<4d. i.e the order s, p, and so on shows that orbitals have lower energy than that of orbital p. The orbitals having lower energy are placed nearer to the nucleus. they do not possess the same energy as each other. It can be seen in the above diagram, orbitals 2s and 2p are not placed at the same levels i.e. The orbitals do not contain the same energies. The important observations revealed from these diagrams are, It is named after German astronomer Walter Grotrian (from the first half of the 20 th century). Energy level diagrams are known as Grotrian diagrams. To study the nature of bonding between the electrons, placement of electrons in orbits and to understand the behavior of elements under certain conditions, energy level diagrams are used.Įnergy level diagrams are the representation of placements or arrangements of orbitals (also known as subshells) according to their increasing energy levels.Ībove is the blank energy level diagram which can be used to represent the electrons for any atom under study. The lower energy level is called the ground state whereas the higher energy levels are known as excited states. When electrons transit from one energy level to another, emission or absorption of energy takes place. This emission of energy is generally in the form of light. On the contrary, when an electron jumps from a higher level to a lower level, they emit energy.
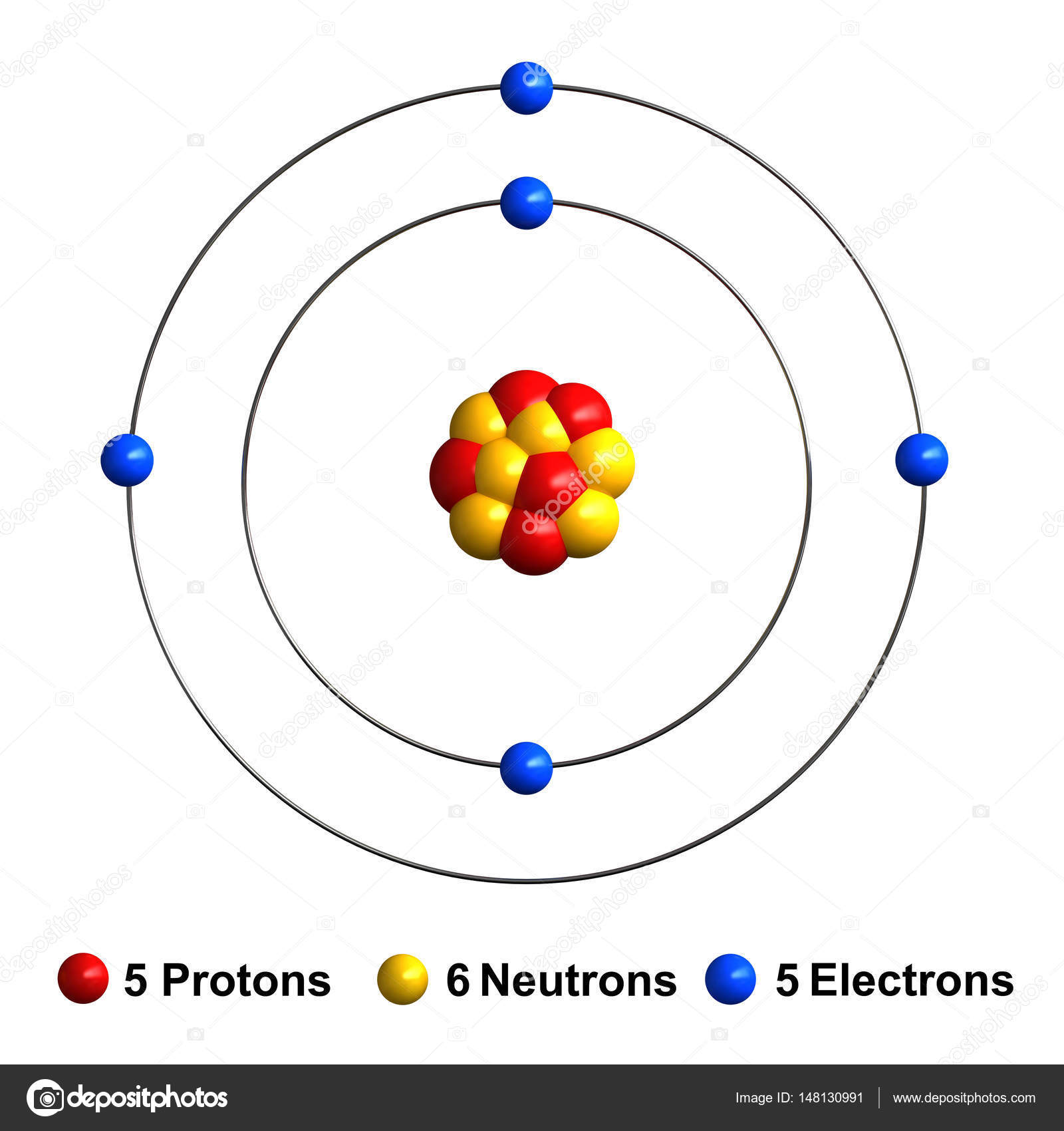
If electrons absorb this fixed energy, it can jump from lower energy level to a higher level. The increase in energy takes place by a fixed amount. Various properties of atoms are based on these valence electrons. The electrons from energy level K contains the least energy whereas the levels that are far from the nucleus contains more energyĮlectrons in the outermost energy level are also called Valence electrons. The second level is called level L, third energy level as M, and so on. The first energy level is also called level 'K'.
/atom--illustration-713786859-5bdb6f7d46e0fb002d6db6df.jpg)
The first four energy levels are shown here. The figure shows the energy levels of an atom. The energy levels are also called electron shells.Īn electron can move in one energy level or to another energy level, but it can not stay in between two energy levels. Their revolution is restricted in particular orbits according to their energy levels.Įnergy levels are nothing but the fixed distances of electrons from the nucleus of an atom. They can’t move freely at any random position. Electrons are small, negatively charged particles that follow a circular path or orbit while moving around the nucleus. An atom consists of electrons, revolving around a nucleus.
